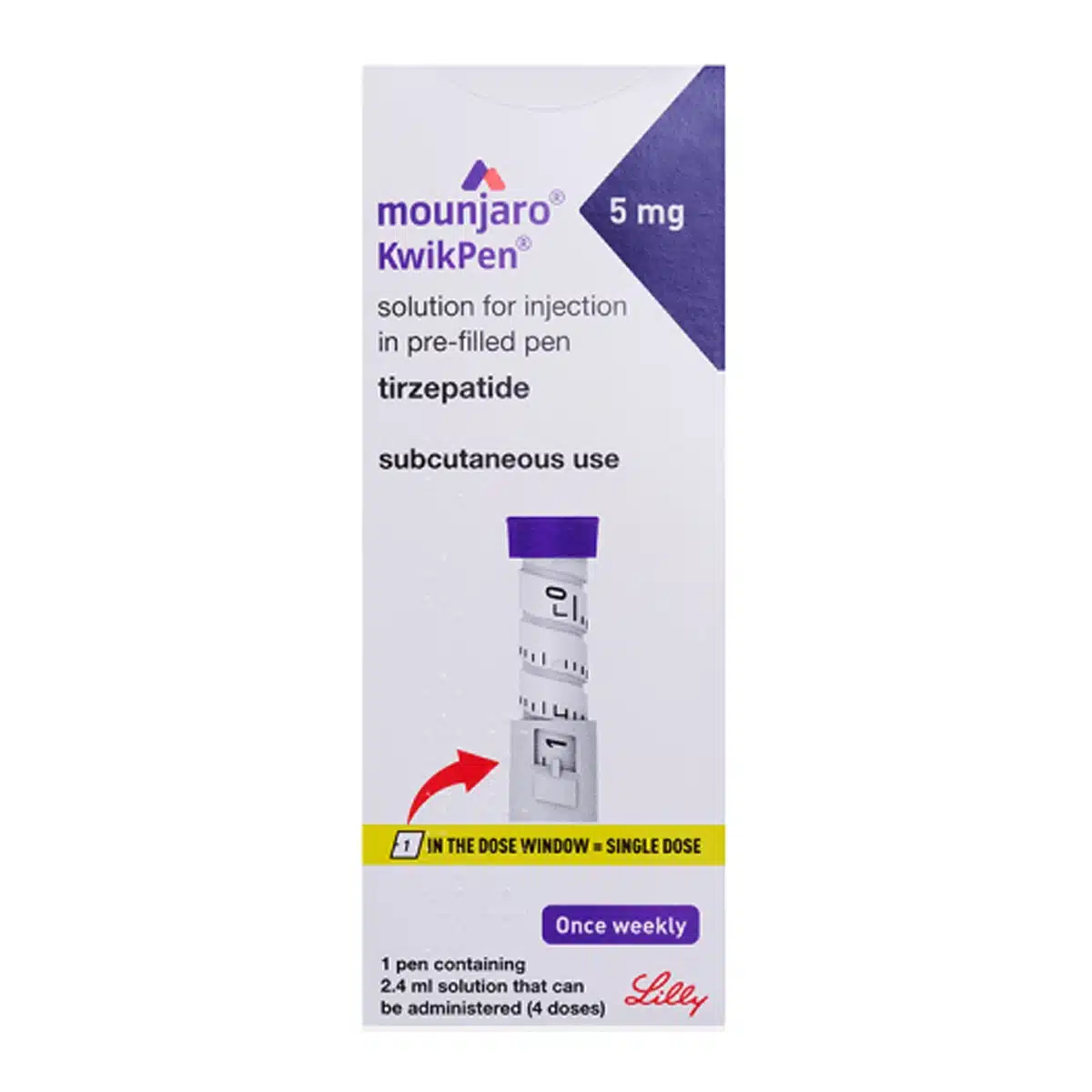
MOUNJARO® 5mg KwikPen®, 1 Stk.
Tier
| Packs | Discount (%) | For Each |
|---|---|---|
| 1 - 3 | — | $499.00 |
| 4 - 10 | 12.02 % | $439.00 |
| 11 - 20 | 14.03 % | $429.00 |
| 21+ | 16.03 % | $419.00 |
Shop with Confidence: Product Authenticity is Guaranteed
All products available at Doctor Medica shop are obtained from respective manufacturers and contain original LOT numbers Contact us if you have any questions about product LOT numbers.
FAQ
MOUNJARO® 5mg KwikPen® is a disposable, multi-dose pre-filled pen that delivers tirzepatide 5 mg in a 0.6 mL subcutaneous injection once weekly. Each KwikPen contains 2.4 mL of solution (total 20 mg tirzepatide; 8.33 mg/mL) and provides 4 fixed doses. It is a dual GIP/GLP-1 receptor agonist used alongside diet and exercise for type 2 diabetes and for weight management in eligible adults. The solution is clear, colourless to slightly yellow and can be injected in the abdomen, thigh, or upper arm.
-
Dose per injection: 5 mg tirzepatide = 0.6 mL
-
Pen capacity: 2.4 mL (total 20 mg), 4 weekly doses
-
Concentration: 8.33 mg/mL
-
Mechanism: GIP and GLP-1 receptor agonist
-
Injection sites: abdomen, thigh, upper arm (rotate sites)
Begin with a once-weekly titration to improve tolerability, then maintain or escalate based on response. Each weekly dose is 0.6 mL (5 mg) delivered subcutaneously with the KwikPen.
-
Starting dose: 2.5 mg once weekly for 4 weeks, then increase to 5 mg once weekly
-
Further titration: Increase in 2.5 mg steps (7.5, 10, 12.5, 15 mg) after ≥4 weeks on the current dose; max 15 mg weekly
-
Weekly timing: Same day each week; may change day if ≥3 days between doses
-
Missed dose: If ≤4 days late, take ASAP; if >4 days, skip and resume on the next scheduled day; never double-dose
-
Technique (KwikPen): Prime the pen, dial to 1 (full 0.6 mL), inject SC, hold 5 seconds, confirm “0” in window before removing
-
Site rotation: Rotate abdomen/thigh/upper arm; different site from any insulin injection
-
Concomitant therapy: Consider reducing sulphonylurea or insulin dose to lower hypoglycaemia risk; monitor glucose
MOUNJARO® (tirzepatide) is indicated for adults with insufficiently controlled type 2 diabetes and for weight management in specified BMI categories. Use is adjunct to diet and exercise.
- Type 2 diabetes:
- Monotherapy when metformin is inappropriate (intolerance/contraindication)
- Add-on to other antidiabetic medicines
- Monotherapy when metformin is inappropriate (intolerance/contraindication)
- Weight management: Adults with BMI ≥30 kg/m², or BMI ≥27 to <30 kg/m² with ≥1 comorbidity (e.g., hypertension, dyslipidaemia, OSA, CVD, prediabetes, T2D)
- Contraindication: Hypersensitivity to tirzepatide or any excipients
- Key cautions: History of pancreatitis, severe GI disease (e.g., severe gastroparesis), certain diabetic retinopathy situations—use with caution and monitor
- Pregnancy/lactation: Not recommended in pregnancy; discontinue ≥1 month before planned pregnancy (long half-life ~5 days). Unknown if excreted in human milk
Common adverse effects (overview): Nausea, diarrhoea, vomiting, abdominal pain, constipation; hypoglycaemia risk increases with sulphonylurea/insulin (see detailed section below).
Each 0.6 mL dose from the KwikPen contains 5 mg tirzepatide in solution. The multi-dose pen contains 20 mg tirzepatide in 2.4 mL (concentration 8.33 mg/mL).
-
Active substance: tirzepatide 5 mg per 0.6 mL dose (KwikPen)Total per pen: 20 mg in 2.4 mL; 4 doses per pen
-
Excipients (not exhaustive): Contains benzyl alcohol ~5.4 mg per 0.6 mL dose (KwikPen)
-
Sodium content: <1 mmol (23 mg) sodium per dose (sodium-free)
-
Appearance: Clear, colourless to slightly yellow solution
The KwikPen is supplied with the medicine’s Patient Information Leaflet (PIL) and Instructions for Use (IFU). Pen needles are not included and must be obtained separately.
-
Device: MOUNJARO® KwikPen multi-dose, single-patient-use pre-filled pen
-
Doses per pen: 4 fixed weekly doses of 0.6 mL each (dial to 1)
-
Documentation: PIL and IFU are included with your medicine
-
Needles: Not included in the pack; use compatible pen needles
Yes—gastrointestinal events are the most frequent and are usually mild to moderate, especially during dose-escalation, and tend to decrease over time. Hypoglycaemia can occur, particularly when used with a sulphonylurea or insulin.
-
GI disorders (T2D trials): Any GI AE in 37.1% (5 mg), 39.6% (10 mg), 43.6% (15 mg) vs 20.4% placebo; nausea 12.2%, diarrhoea 11.8% at 5 mg; discontinuation due to GI events 3.0% (5 mg) vs 0.4% placebo
-
Weight-management trials (no T2D): GI disorders 55.6–60.8% on tirzepatide vs 30.3% placebo; mostly mild/moderate
-
Hypoglycaemia (T2D): With sulphonylurea: 10–14%; with basal insulin: 14–19% (clinically significant <3.0 mmol/L); severe events overall 0.2% of patients
-
Injection-site reactions: 3.2% (T2D) and 8.0–8.6% (weight mgmt/OSA) vs 0.4–2.6% placebo
-
Gallbladder events: Acute disease reported in up to 2.0% on tirzepatide vs up to 1.6% placebo; association with weight reduction observed
-
Pancreatitis: Acute pancreatitis reported (uncommon); discontinue if suspected
-
Cardiac conduction/HR: Mean +3–5 bpm increase; small PR increases; arrhythmia incidence similar to placebo
-
Lab changes: Mean amylase +23–38%, lipase +31–42% vs minimal placebo change
-
Hypersensitivity: Reported; anaphylaxis/angioedema rare
Store unused pens refrigerated and do not freeze. After first use, pens can be kept at room temperature for a limited time and must be discarded on schedule.
-
Unused pens: Refrigerate at 2–8 °C; do not freeze; if frozen, do not use
-
After first use: May store at ≤30 °C; keep away from heat; discard 30 days after first use
-
Doses per pen: Dispose after 4 weekly doses; some solution remains by design—do not attempt extra dosing
-
Needle handling: Do not store with needle attached; remove and dispose in a sharps container after each dose
-
Safety: Keep out of sight and reach of children