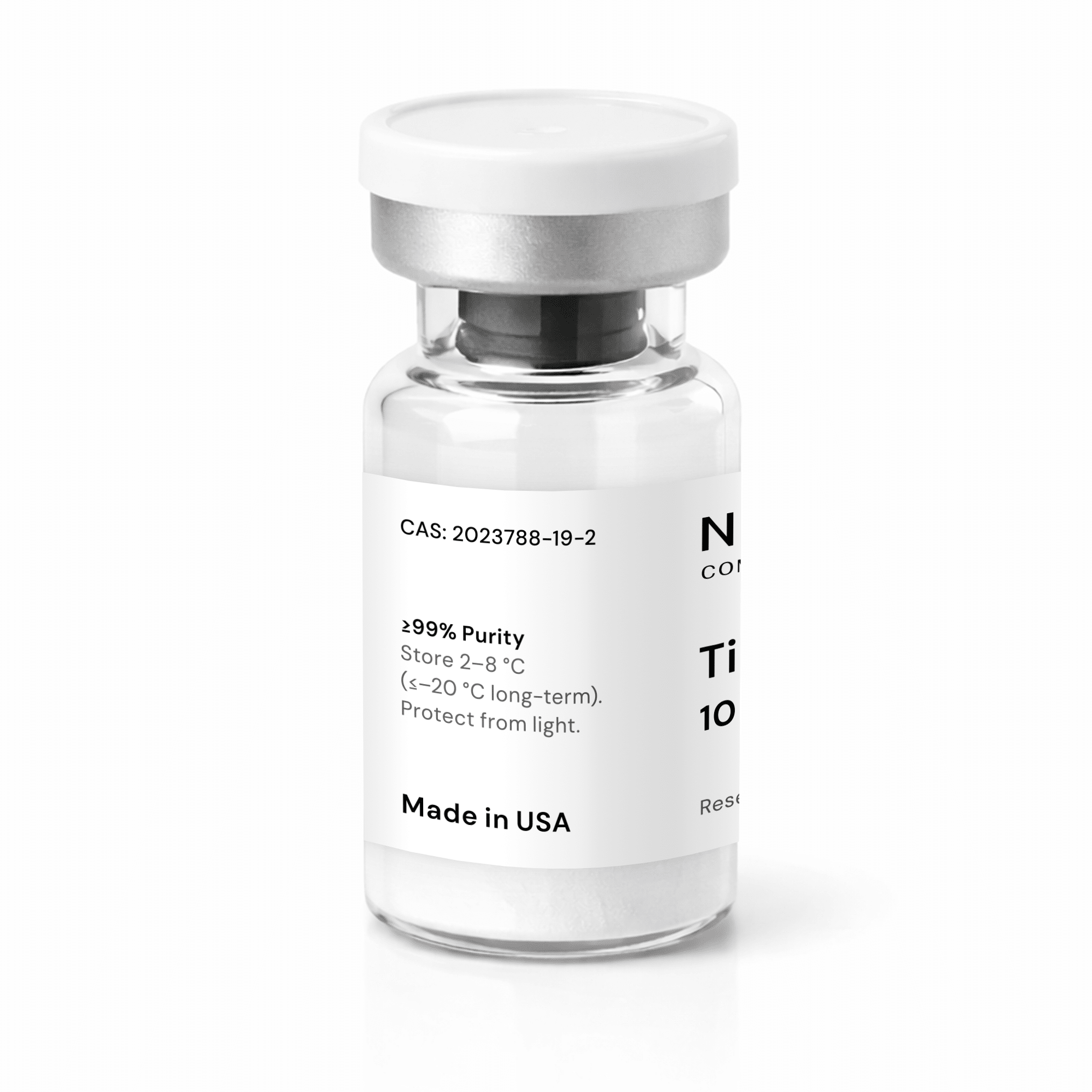Tirzepatide 10mg
Disclaimer
DISCLAIMER: The product is delivered in powdered (lyophilized) form and must be properly reconstituted prior to research use..
Research Use Only:
This product is strictly for laboratory experimentation and in vitro testing. Human administration is prohibited. All information regarding Novera Compounds available on the Doctor Medica website is for educational purposes only and is intended for use by licensed, qualified professionals.
Tier
| Packs | Discount (%) | For Each |
|---|---|---|
| 1 - 5 | — | $170.00 |
| 6 - 10 | 10 % | $153.00 |
| 11 - 20 | 18.82 % | $138.00 |
| 21+ | 27.06 % | $124.00 |
Product description
Tirzepatide 10mg - About This Product
Tirzepatide 10mg, also known as P1206, LY3298176, tirzepatida, tirzepatidum, OYN3CCI6QE, is a synthetic 39–amino acid peptide developed for advanced metabolic and endocrine research. It is engineered as a tirzepatide dual agonist, meaning it has been designed to interact with both glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) biological systems. The peptide sequence is based on native GIP and includes specific structural modifications intended to enhance molecular stability and extend circulation time in research models. These modifications include the incorporation of aminoisobutyric acid (Aib) residues and conjugation of a C20 fatty diacid to a lysine residue via a hydrophilic linker, which increases albumin binding. Tirzepatide peptide has a molecular weight of approximately 4810.5 g/mol and is supplied as a lyophilized white to off-white powder with ≥98% purity, verified by analytical testing such as HPLC/MS. The 10mg vial format is commonly used in dose-escalation and mid-to-higher dose research protocols investigating metabolic regulation, energy balance, and gastrointestinal signaling.- Chemical Formula: C₂₂₅H₃₄₈N₄₈O₆₈
- Peptide Sequence: Tyr-Aib-Glu-Gly-Thr-Phe-Thr-Ser-Asp-Tyr-Ser-Ile-Aib-Leu-Asp-Lys(C20-linker)-Ile-Ala-Gln-Lys-Ala-Phe-Val-Gln-Trp-Leu-Ile-Ala-Gly-Gly-Pro-Ser-Ser-Gly-Ala-Pro-Pro-Pro-Ser-NH₂
- CAS Number: 2023788-19-2
Tirzepatide 10mg - Key Features and Benefits
- Research-grade purity of ≥98%, verified by HPLC/MS for consistency and reproducibility
- Supplied as a lyophilized powder in sealed vials for controlled laboratory handling
- Designed for studies exploring combined incretin signaling, including investigators who buy tirzepatide for non-clinical research use
- Structural modifications support extended stability and prolonged biological exposure in experimental models
- Suitable for mid-range and advanced dose-escalation studies using the tirzepatide 10 mg peptide format
- Used in research examining metabolic, gastrointestinal, and neuroendocrine systems
- Stable under recommended refrigerated storage conditions when handled correctly
- For laboratory research use only, clearly labeled for compliant, non-therapeutic use
Tirzepatide 10mg - Mechanism & Research Applications
Tirzepatide has been investigated as a single-molecule dual incretin agonist, producing broader metabolic effects than agents targeting only one incretin pathway. In research settings, studies examining tirzepatide for research have focused on the following areas:- Glucose Regulation: Studied for its role in enhancing glucose-dependent insulin secretion and moderating glucagon release in experimental models
- Energy Balance and Appetite: Investigated for effects on central and peripheral signaling involved in satiety, caloric intake, and energy expenditure
- Lipid Handling: Evaluated for dose-dependent effects on postprandial lipid metabolism and adipose tissue signaling
- Gastrointestinal Function: Examined for its influence on gastric emptying rates and nutrient absorption timing
- Metabolic Flexibility: Explored for its ability to influence substrate utilization and overall energy homeostasis
Tirzepatide 10mg - Dosing & Observed Effects in Research
In research contexts, tirzepatide has been evaluated using structured dose-escalation protocols to characterize pharmacokinetics and biological response patterns. In human clinical research programs, dosing commonly begins at 2.5 mg once weekly and increases in 2.5 mg increments every four weeks. The 10mg dose represents an intermediate-to-higher level within this escalation framework and is frequently used to assess dose–response relationships. Furthermore, published clinical trial programs, including large-scale metabolic studies, report that tirzepatide 10mg is associated with measurable, dose-dependent changes in glucose handling, insulin secretion dynamics, lipid parameters, and appetite-related biomarkers. Pharmacokinetic data indicate a time to peak concentration ranging from approximately 8 to 72 hours following subcutaneous administration, with an estimated half-life of around five days, supporting once-weekly dosing in research protocols. Additionally, gastrointestinal observations, such as nausea or delayed gastric emptying, have been reported more frequently at higher doses, including 10mg, within controlled research settings.Tirzepatide 10mg - Storage, Safety & References
- Unreconstituted: Tirzepatide 10mg should be stored as a lyophilized powder in its original sealed vial at 2–8°C, protected from light and moisture to maintain stability. Refrigerated storage is recommended prior to reconstitution, and room-temperature storage is only acceptable during transport.
- Reconstituted: Once reconstituted using sterile diluent, solutions should be kept refrigerated at 2–8°C and used within the timeframe specified by the research protocol, typically within 28–30 days. Avoid freeze-thaw cycles, as they can degrade the peptide's quality.
- Reconstitution: Reconstitution should be performed using aseptic technique with sterile equipment. The vial should be gently swirled rather than shaken to avoid peptide aggregation.
- Aseptic handling: Standard laboratory personal protective equipment, including gloves, eye protection, and a lab coat, should be used during handling
References
- Willard FS, Douros JD, Gabe MB, et al. Tirzepatide is an imbalanced and biased dual GIP and GLP-1 receptor agonist. JCI Insight. 2020;5(17):e140532. Published 2020 Sep 3. doi:10.1172/jci.insight.140532
- Fisman EZ, Tenenbaum A. The dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist tirzepatide: a novel cardiometabolic therapeutic prospect. Cardiovasc Diabetol. 2021;20(1):225. Published 2021 Nov 24. doi:10.1186/s12933-021-01412-5
- Coskun T, Sloop KW, Loghin C, et al. LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus: From discovery to clinical proof of concept. Mol Metab. 2018;18:3-14. doi:10.1016/j.molmet.2018.09.009
Compliance Notice
This product is intended for laboratory research use only and is not approved for human or veterinary use.Shop with Confidence: Product Authenticity is Guaranteed
All products available at Doctor Medica shop are obtained from respective manufacturers and contain original LOT numbers Contact us if you have any questions about product LOT numbers.